Sales Force Automation in the Pharmaceutical Industry: What Actually Matters in 2026
Your Veeva renewal lands, and buried in the paperwork is a quiet phrase that changes your next two years: "Vault CRM migration." At the same time, finance wants a straight answer on why you're paying enterprise prices for something reps treat like a call log with extra steps.
Sales force automation in the pharmaceutical industry is growing fast, but the growth isn't the point. The point is that the rules of the road have changed: the Veeva-Salesforce split is real, timelines are tight, and the teams that win in 2026 are the ones that stop treating CRM as "the system" and start treating it as one piece of a commercial operating model.
Let's break this down the way we'd do it in a steering committee: what to decide, what to ignore, and what'll blow up your timeline if you pretend it's "phase two."
What you need now
If you're on Veeva CRM (Salesforce), the end-of-support date is September 2030. That sounds far away until you map it to reality: global pharma CRM programs routinely take 18-36 months once you count affiliate design, data harmonization, validation, integrations, and field adoption. Start late and you're not "behind schedule" - you're migrating under a hard deadline with no room for surprises.
Your practical shortlist usually ends up here:
- Veeva Vault CRM
- Salesforce Life Sciences Cloud
- Microsoft Dynamics 365 (especially for mid-market)
And here's the thing that frustrates us every time: teams spend months arguing about screens and workflows, then try to "clean up the data later." Later never comes. If your HCP data's messy, your SFA will look broken even if the implementation is technically perfect.
What pharma SFA actually does
Pharma SFA isn't generic CRM with a healthcare theme. It's built around four workflows that define a rep's day, and each one has compliance and audit expectations baked in.
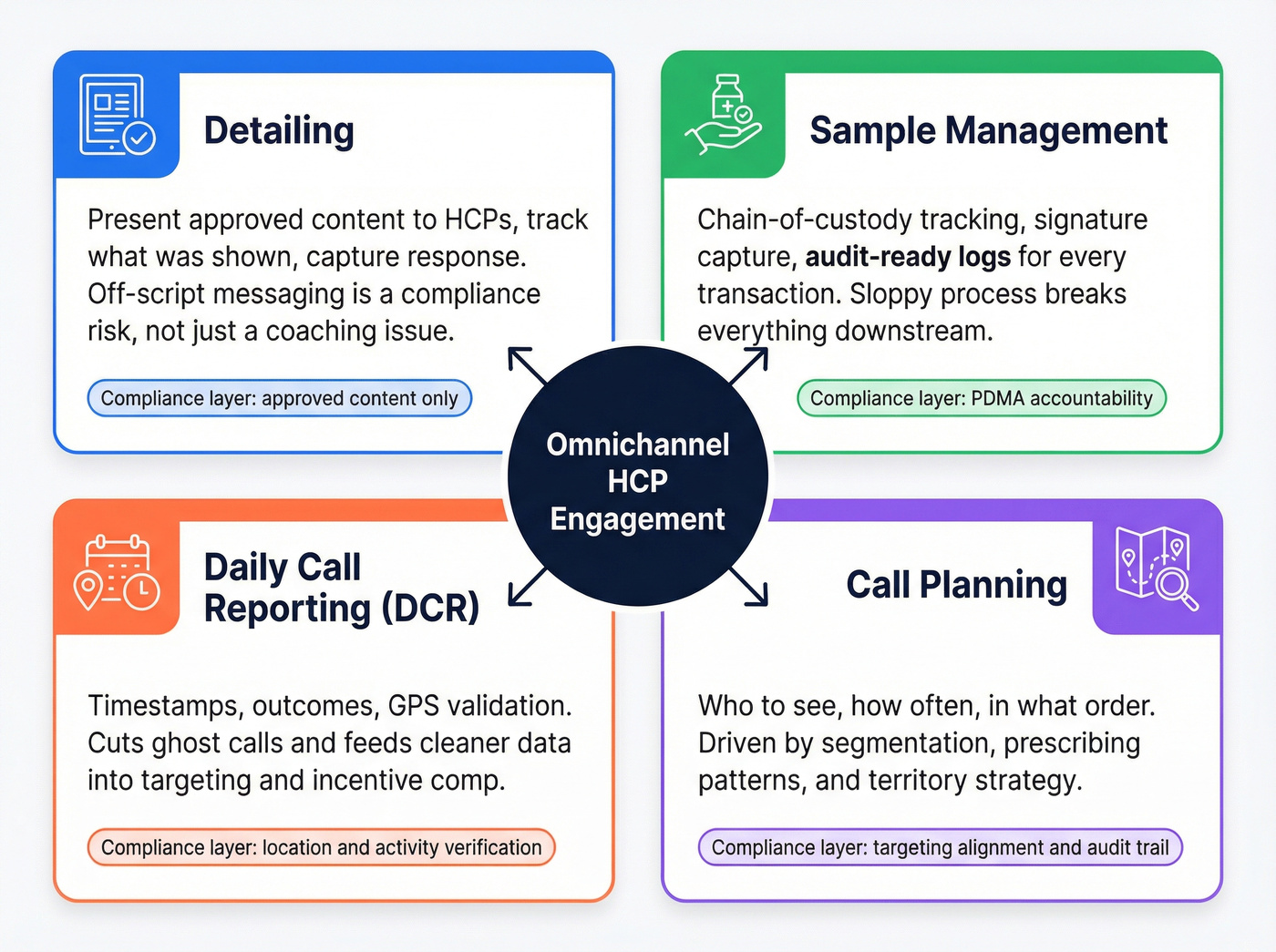
Detailing is controlled messaging: presenting approved content, tracking what was shown, and capturing the HCP response. The guardrails matter because "off-script" isn't just a coaching issue in pharma - it's a risk issue.
Sample management is accountability: chain-of-custody tracking, signature capture, and audit-ready logs for every transaction. If your process is sloppy, it won't matter how pretty the UI is.
Daily Call Reporting (DCR) is the record: timestamps, outcomes, and often location validation. GPS stamping exists for a reason: it cuts down on "ghost calls" and forces cleaner activity data, which then feeds targeting and incentive comp.
Call planning is the math: who to see, how often, and in what order based on segmentation, prescribing patterns, and territory strategy.
The scope's also expanding. Modern pharma SFA increasingly ties in omnichannel HCP engagement so field activity doesn't fight digital activity. Most generic CRMs can be configured to approximate this, but you pay for it in customization, validation work, and ongoing admin overhead.
Why this matters in 2026
The Veeva-Salesforce split is the biggest commercial-tech forcing function pharma has seen in years. Veeva won't renew its Salesforce platform agreement, and Veeva CRM on Salesforce is in stability mode: security patches and critical fixes, not meaningful innovation.
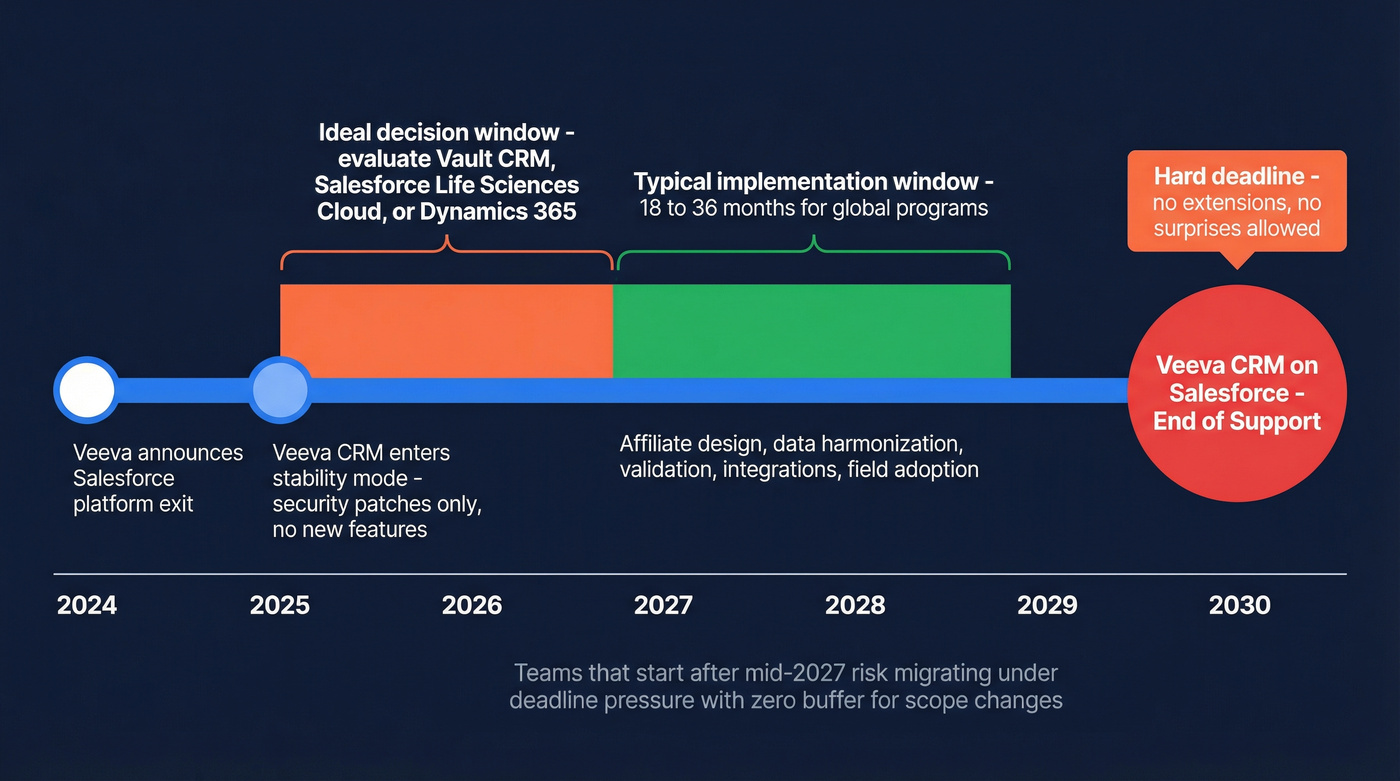
That changes how you should evaluate vendors. You're not just buying features; you're buying a path for the next decade of your commercial stack, plus the ability to hire and retain people who can run it.
AI is also raising expectations inside leadership teams. Commercial leaders are no longer satisfied with "we have a CRM." They want proof that the platform improves targeting, reduces wasted effort, and helps reps spend more time with the right HCPs. If your SFA can't support that story with clean data and measurable workflows, it becomes a budget target.
For context and primary references on the regulatory side, start with FDA 21 CFR Part 11 guidance and the CMS Open Payments program:
- https://www.ecfr.gov/current/title-21/chapter-I/subchapter-A/part-11
- https://openpaymentsdata.cms.gov/ And for samples, the PDMA framework is the anchor:
- https://www.fda.gov/drugs/drug-supply-chain-security-act-dscsa/prescription-drug-marketing-act-pdma

SFA rollouts fail when rep lists are wrong. Prospeo enriches HCP/account records with 98% verified emails and 125M+ verified mobiles - refreshed every 7 days to keep territories current.
Start with clean contact data - then automate the workflow.
Vendor landscape (what's actually different)
Here's the honest version: at the enterprise end, you're mostly choosing an ecosystem and an operating model. At the mid-market end, you're choosing how much complexity you can afford to carry.
If you need a quick gut-check on what "CRM" can mean across industries, skim a few examples of a CRM before you lock requirements.
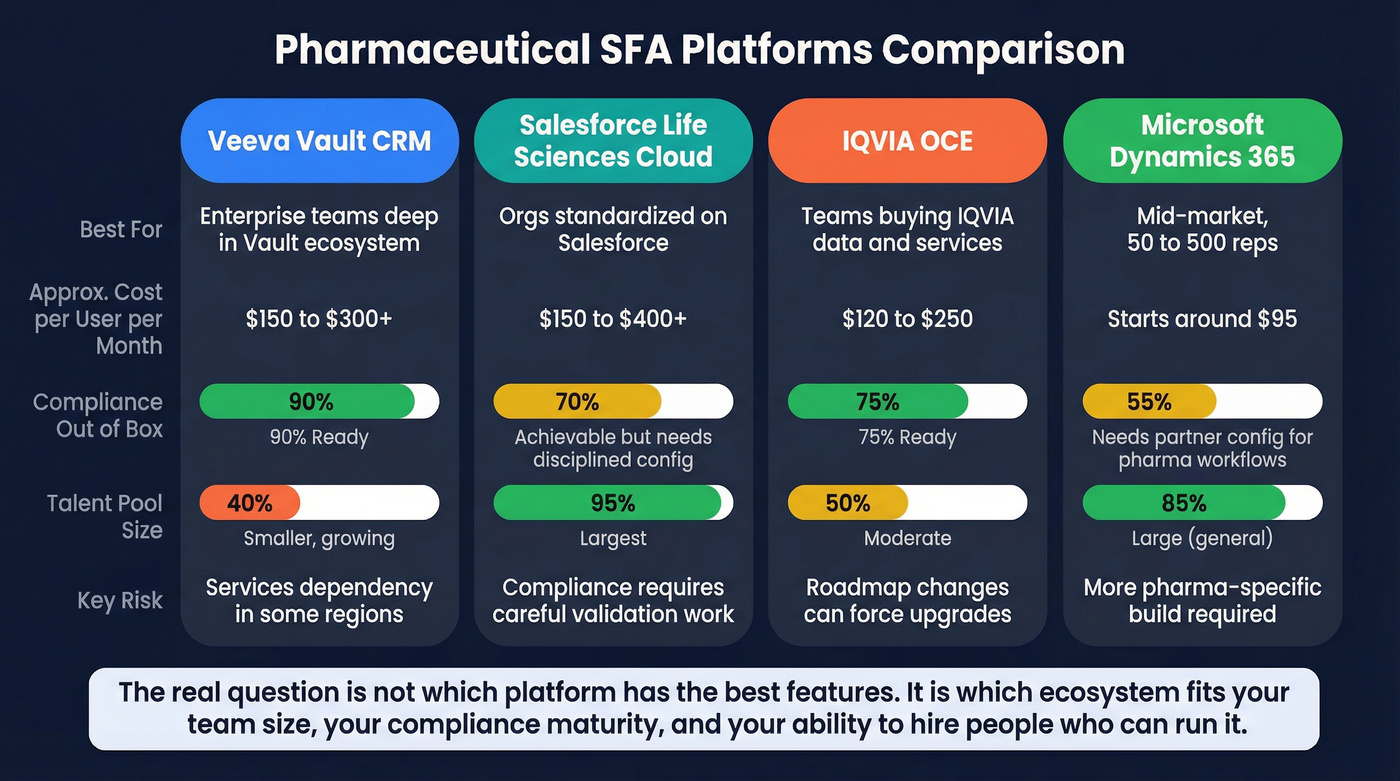
| Platform | Best for | Strengths | Key risk | Approx. cost |
|---|---|---|---|---|
| Veeva Vault CRM | Enterprise teams already deep in Veeva | Tight Vault alignment, strong compliance posture, X-Pages for codeless admin | Smaller talent pool and higher services dependency in some regions | $150-300+/user/mo + $500K-5M+ implementation |
| Salesforce Life Sciences Cloud | Orgs standardized on Salesforce | Large talent pool, strong platform ecosystem, AI features for rep productivity | Compliance is achievable but takes disciplined configuration + validation | $150-400+/user/mo + implementation |
| IQVIA OCE | Teams that want CRM tied closely to IQVIA data | Strong fit if you're already buying IQVIA data + services, solid commercial suite | Roadmap and support timelines can drive forced change later | ~$120-250/user/mo (often custom) |
| Microsoft Dynamics 365 | Mid-market and cost-sensitive rollouts | Lower total cost for many teams, flexible platform, easier licensing story | More pharma-specific workflows require partner build/config | Starts around ~$95/user/mo (varies by edition) |
A quick scenario from a program we watched up close: a regional affiliate insisted on keeping its "unique" territory structure and local HCP lists. Six months later, the global template was ready, but the affiliate couldn't migrate because their HCP IDs didn't match anything upstream, and the integration team had to build a one-off mapping layer that nobody wanted to own. That single decision added months, not weeks.
Veeva Vault CRM: best when Vault is already your center of gravity
If your content, compliance, and medical teams already live in Vault, Vault CRM is the cleanest path. You avoid turning your migration into a multi-system replatform, and you keep governance in one place.
X-Pages is also a real advantage for day-to-day operations. In our experience, teams that rely on codeless admin for common changes (layouts, flows, content presentation) move faster and spend less time in the "submit a ticket, wait two sprints" loop. That speed matters once the field starts asking for changes every week.
Skip Vault CRM if you're not ready to commit to the broader Veeva ecosystem. Buying it "just for CRM" while the rest of your stack lives elsewhere is how you end up paying premium prices for integration work that never stops.
Salesforce Life Sciences Cloud: best when Salesforce is your platform standard
If your IT org is already built around Salesforce, Life Sciences Cloud keeps your talent pool, your platform governance, and your integration patterns consistent. That's not a soft benefit; it's the difference between hiring a team in three months versus nine.
Where Salesforce teams get burned is assuming compliance is automatic. The platform supports audit trails and controls, but you still have to design, configure, validate, and document your specific processes. If you treat that as an afterthought, you'll pay for it twice: once in rework, and again in credibility with QA.
If you’re pressure-testing license and admin overhead, it helps to compare against other contact management software to separate “must-have” from “nice-to-have.”
Microsoft Dynamics 365: the mid-market option people ignore (and shouldn't)
Here's our slightly opinionated take: if you're a 50-500 rep organization, you should at least price and pilot Dynamics 365 before defaulting to Veeva or Salesforce.
Real talk: a lot of mid-market teams don't use the advanced features they're paying for. They need reliable call planning, clean activity capture, basic segmentation, and integrations that don't collapse every quarter. Dynamics can cover that, and the total cost difference is often big enough to fund the data work and change management that actually drives adoption.
On Reddit threads (especially in r/CRM and adjacent sales ops communities), the recurring theme is the same: "We bought the enterprise thing, then used 20% of it." That isn't a moral failing. It's just what happens when procurement optimizes for brand safety instead of fit.
Compliance requirements (and the part vendors don't do for you)
Any pharma SFA platform has to support the basics, but "the software can do it" isn't the same as "we're compliant."
- 21 CFR Part 11: audit trails, electronic signatures tied to individual users, access controls, and validation documentation.
- PDMA sample accountability: chain-of-custody records, signature capture, and audit-ready transaction logs.
- Sunshine Act / Open Payments: tracking and reporting transfers of value to HCPs.
Budget for validation and documentation like it's a first-class workstream, because it is. We've seen teams spend more on validation support than they expected to spend on licenses, and it wasn't because anyone was wasteful - it was because they under-scoped the work.
If you want a practical way to operationalize this, treat it like sales process optimization: define the workflow, define the evidence, then automate.
The upstream problem: HCP data quality
Most pharma SFA conversations obsess over platform features. That's backwards.
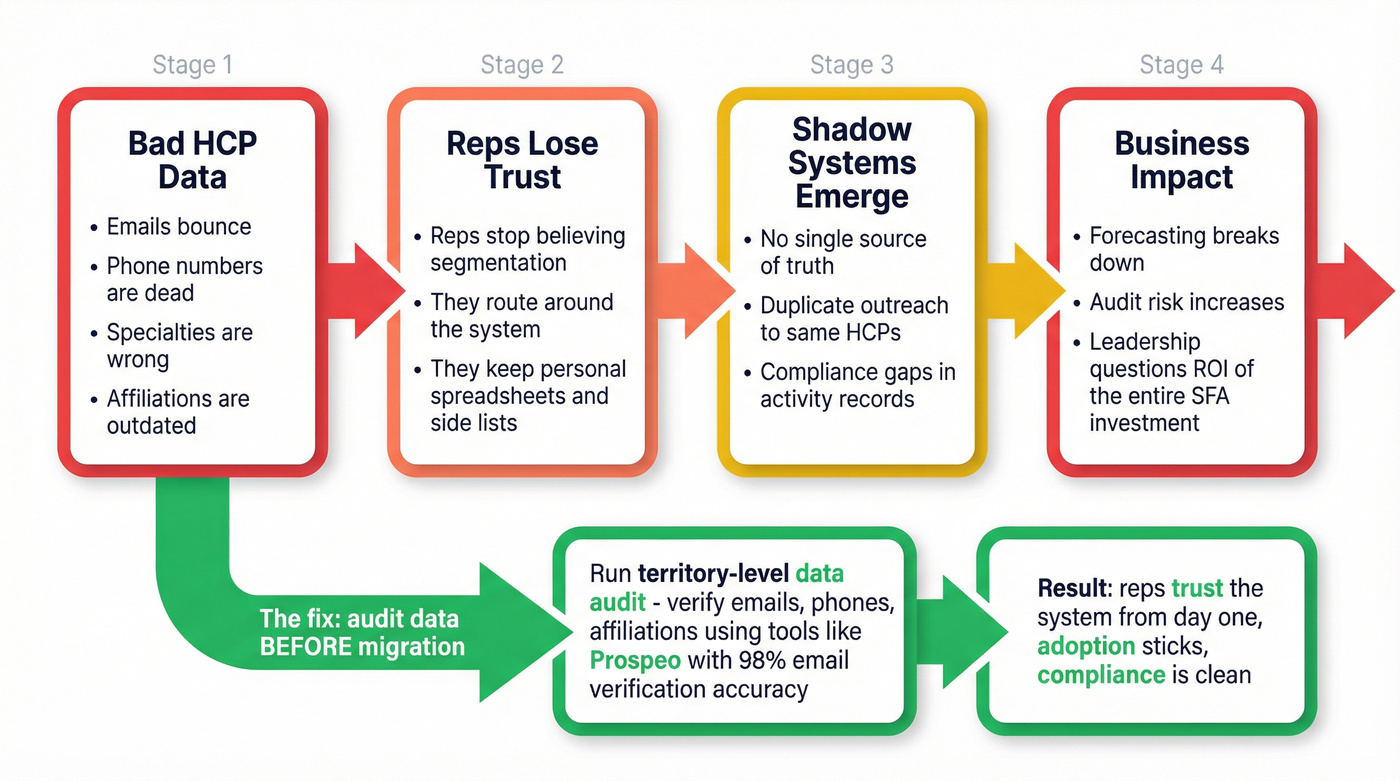
If your HCP emails bounce, phone numbers are dead, specialties are wrong, and affiliations are outdated, reps will route around the system. They'll keep their own lists, they'll stop trusting segmentation, and your "single source of truth" becomes a compliance risk and a forecasting problem at the same time.
One of the fastest ways to de-risk an SFA program is to run a data audit before you migrate. Not after. Before. Pick a few territories, pull the HCP list you think is "good," and measure how much of it is actually usable for outreach and follow-up.
This is also where tools like Prospeo fit naturally: you can upload an existing contact list for email verification, spot obvious decay, and fill gaps using a database of 300M+ professional profiles refreshed every 7 days. Prospeo verifies emails in real time with 98% accuracy, and the free tier (75 verified emails per month) is enough to sanity-check a territory without turning it into a procurement project.
If you’re comparing vendors for this layer, start with a shortlist of data enrichment services and work backward from your required match rate and refresh cadence.

Migrating from Veeva CRM to Vault or Life Sciences Cloud? Stop building mapping layers around stale IDs. Use Prospeo’s CRM/CSV enrichment (83% match rate, 50+ data points) to standardize records fast.
De-risk migration timelines with weekly-refreshed commercial data.
Implementation realities (timelines people regret ignoring)
Single-market deployments often land in 6-12 months if scope is controlled and integrations are reasonable. Global transformations are a different animal: multiple affiliates, template governance, data harmonization, integration rebuilds, full validation documentation, and field change management. Plan on 18-36 months, and expect $500K-5M+ in implementation costs depending on scale and complexity.
If you need a way to keep leadership aligned on outcomes, borrow a page from sales operations metrics: define adoption, data quality, and cycle-time KPIs before build starts.
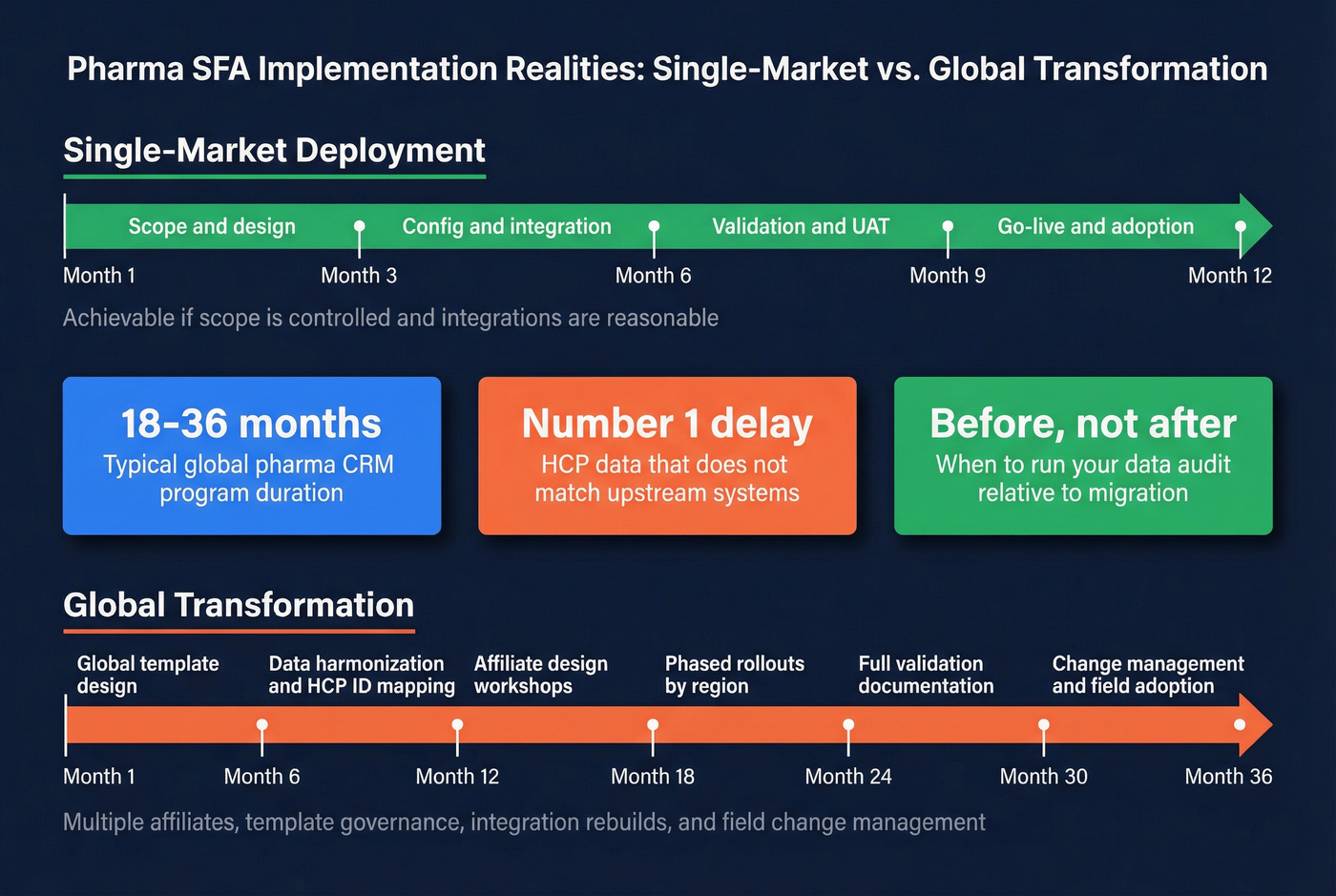
The repeat offenders we keep seeing:
- Affiliate data harmonization takes twice as long as planned. Everyone agrees in principle, then fights over definitions for weeks.
- Validation work eats the budget. Not because it's optional, but because it's tedious, detailed, and unavoidable.
- Change management gets pushed to the end. Then go-live week turns into a fire drill, and adoption drops right when leadership expects a win.
Build buffer into the plan. You'll use it.
FAQ
What's the difference between pharma SFA and generic CRM?
Pharma SFA includes sample management with PDMA accountability, DCR with audit-ready activity capture (often including location validation), HCP detailing with approved messaging controls, and support for 21 CFR Part 11 requirements. Generic CRM doesn't ship with those workflows ready to run.
How long does a pharma SFA implementation take?
A single-market rollout usually takes 6-12 months. A global, multi-affiliate program with integrations and full validation documentation typically takes 18-36 months, and data harmonization is the most common schedule killer.
How do we make sure our SFA contact data is accurate?
Treat data quality as an upstream workstream, not a cleanup task. Audit a few territories first, verify emails before they enter the CRM, and set a refresh cadence you can actually maintain. Tools like Prospeo help by verifying emails in real time (98% accuracy) and refreshing records every 7 days, so your "good" list doesn't decay for months before anyone notices.
Should mid-market pharma companies choose Veeva or Salesforce?
Not automatically. For many 50-500 rep teams, Microsoft Dynamics 365 can deliver better ROI because it covers the core workflows without the same license and services overhead. The right answer depends on ecosystem fit, compliance workload, and how much of the enterprise feature set you'll truly use.