Biotechnology Email Outreach: What Actually Works in 2026
Your CEO saw a competitor land a CDMO partnership and wants you to "do some outreach to biotechs." Simple enough, right? Except biotechnology email outreach plays by different rules than anything in SaaS. Sales cycles run 6-18 months with up to nine stakeholders - procurement, CMC leads, regulatory, quality, and the CSO who won't reply to anyone below VP level. Layer on FDA and EMA regulatory scrutiny, and every email you send into a pharma inbox either builds credibility or permanently burns it. Generic outreach doesn't just fail here. It gets your domain blacklisted by the exact people you need to reach next quarter.
Quick-Start Checklist
Biotech cold email averages a 3.58% reply rate, but only 14.1% of those replies are positive. That's roughly 5 interested responses per 1,000 emails sent. Here's how to beat the average:
- Keep emails 40-60 words. Decision fatigue is real in biotech.
- Reference biotech-specific triggers - IND filings, funding rounds, clinical milestones.
- Verify every email before sending. Bounces kill your domain faster than bad copy kills your reply rate. (If you need a deeper playbook, start with email deliverability.)
- Warm your domain for 6-8 weeks before scaling volume. Three weeks isn't enough anymore - especially if you’re not tracking email velocity.
Benchmarks That Actually Matter
Instantly's 2026 benchmark report analyzed billions of cold email interactions and found a 3.43% average reply rate across all industries. Top-quartile campaigns hit 5.5%+, and the elite top 10% reached 10.7%+. Biotech outperforms the overall average of 2.09% with that 3.58% reply rate, based on a dataset of 2M+ emails. If you want more context on what “good” looks like across outbound, compare against broader cold email marketing benchmarks.
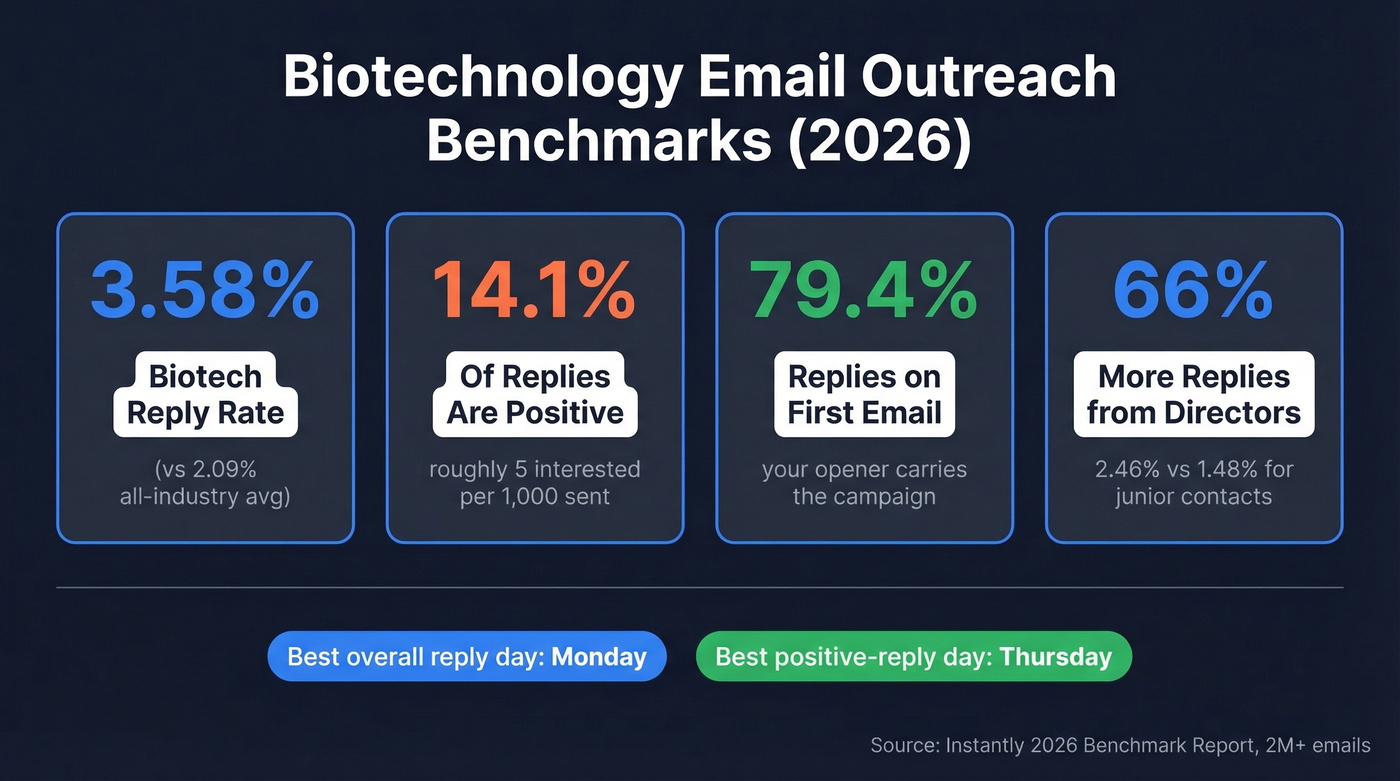
Two targeting insights worth internalizing. Directors reply 66% more than junior contacts - 2.46% vs 1.48%. In our experience, this is the single highest-ROI targeting change most biotech outreach teams can make. (This is also where sales prospecting techniques like role-based segmentation pay off.) And 79.4% of all replies land on the first email in your sequence, so your opener carries the campaign.
Timing matters too. Monday has the highest overall reply rate, while Thursday has the highest positive-reply rate. If you care about "interested" replies specifically, Thursday is the move - then iterate using a best time to send cold emails testing cadence.
Writing Biotech Emails That Get Read
The consensus on r/copywriting is clear: 40-60 words with a value-first structure. Subject lines should be 1-5 words. No fake reply chains, no emoji, no "Quick question." Skip subject lines with the recipient's first name - it signals automation immediately. Reference a program name or a colleague instead. If you need a swipe file, pull from these email subject line examples.
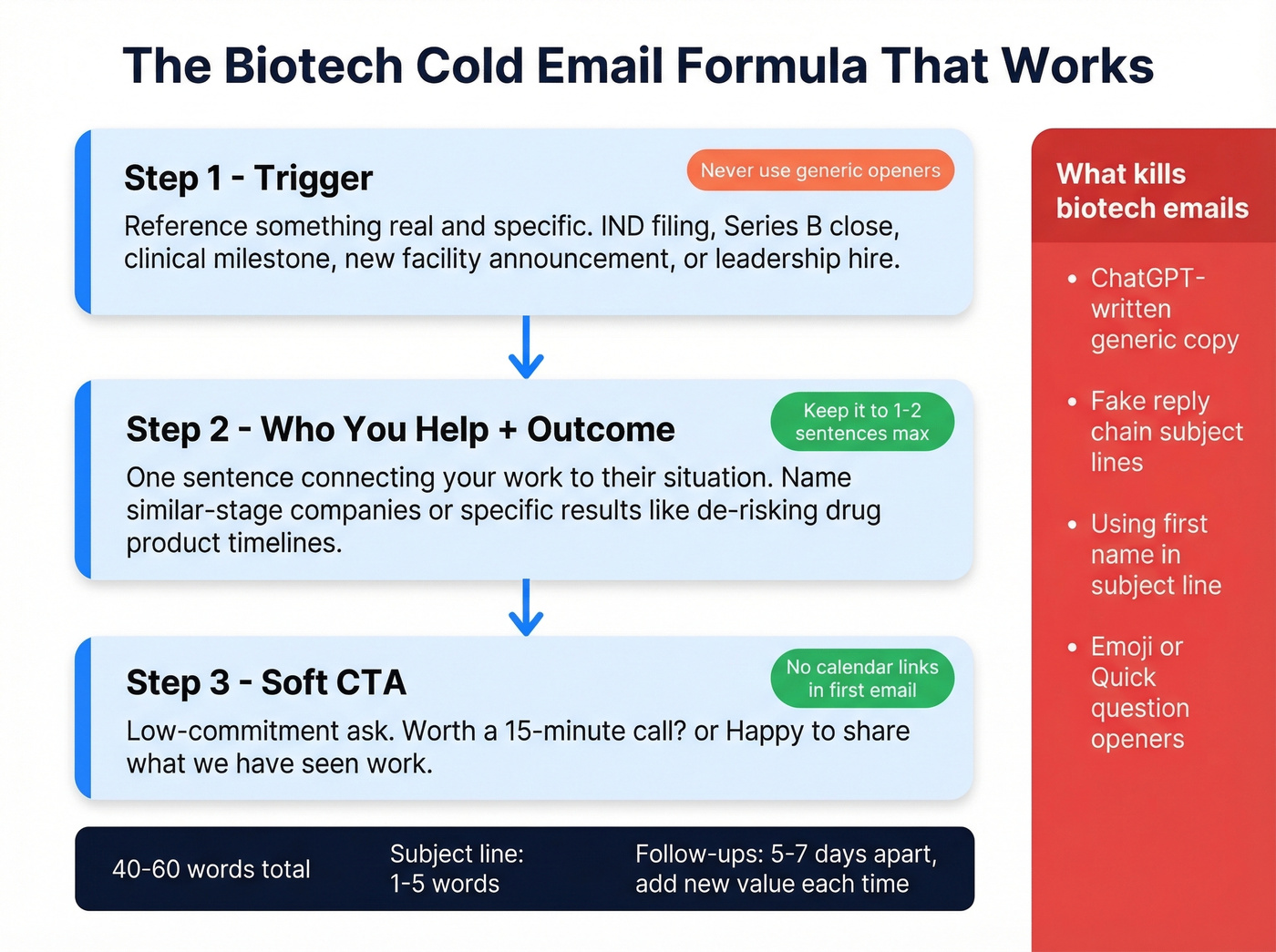
ChatGPT-written biotech emails fail for a specific reason: they can't reference real IND filings or clinical milestones because they don't know the prospect's pipeline. The structure that works is trigger, then who you help plus outcome, then soft CTA. Reference an IND filing, a Series B close, a clinical milestone, or a new facility announcement. (For more on using AI without sounding generic, see AI cold email outreach.)
Two templates that work:
Subject: IND timeline
Hi [Name],
Saw [Company] filed the IND for [program] - congrats. The scale-up and tech transfer timeline from here usually gets tight fast.
We've helped 3 similar-stage sponsors de-risk the drug product side without slipping their CTA date. Worth a 15-minute call to compare notes?
Subject: Series B next steps
Hi [Name],
Congrats on the raise. Most teams at your stage start scaling CMC before they've locked a CDMO - and that's where timelines slip.
Happy to share what we've seen work. 15 minutes?
Space follow-ups 5-7 days apart, with each adding new value rather than just bumping the thread. If you want plug-and-play options, adapt these cold email follow-up templates.

You're sending 1,000 emails to get 5 interested biotech prospects. Every bounce eats into that math. Prospeo's dedicated biotechnology email list delivers 98% verified accuracy with a 7-day refresh cycle - so your IND-filing triggers hit real inboxes, not dead addresses.
Stop burning biotech domains on stale data. Verify before you send.
Fix Your Email Infrastructure First
Here's the thing: most biotech outreach fails before the prospect ever reads a word. It fails at the infrastructure layer.
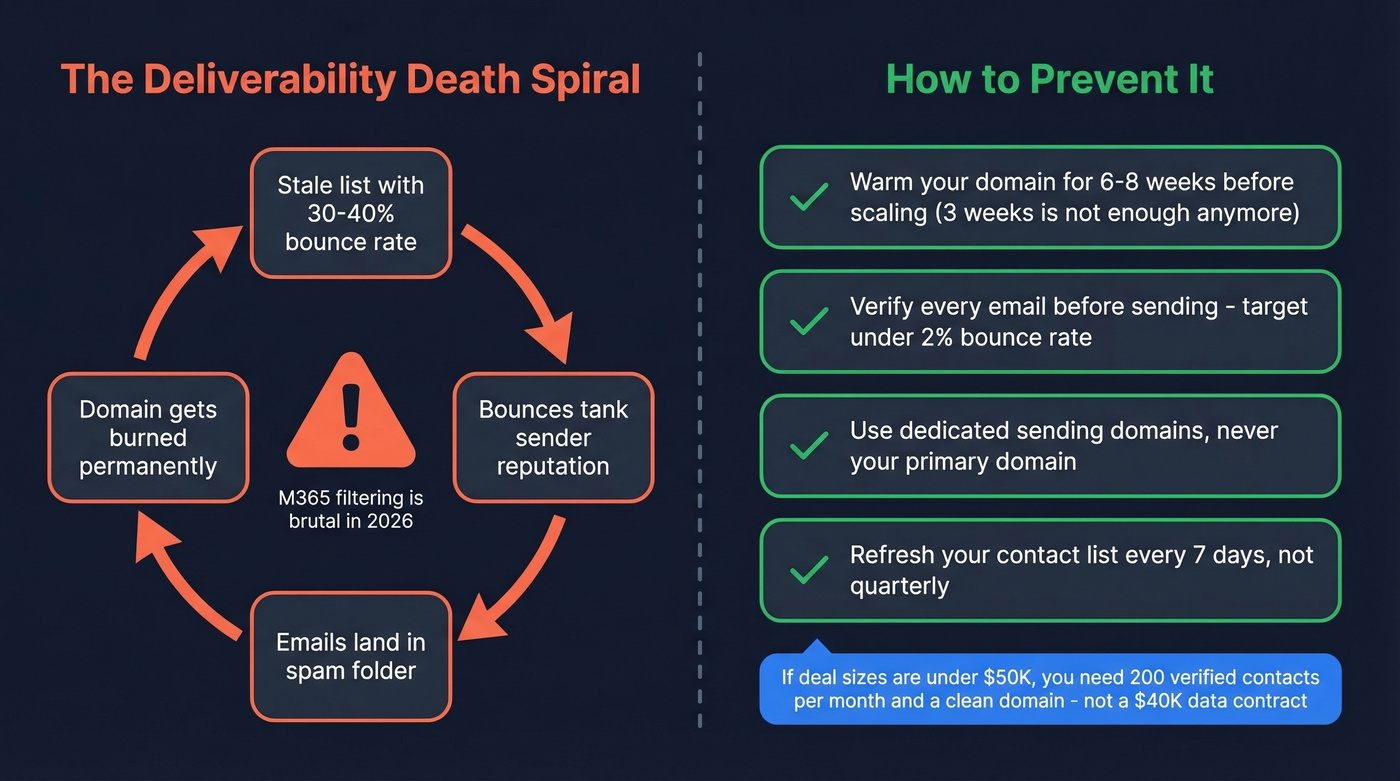
Microsoft 365 filtering has gotten brutal - practitioners on Reddit call M365 "a killer" for cold email deliverability. Domain warm-up that used to take three weeks now takes 6-8 weeks. And if you're sending to stale lists and seeing 30-40% bounce rates, you're accelerating a death spiral: bounces tank your reputation, your reputation lands you in spam, and then the domain is burned. If you’re diagnosing this, start with email bounce rate and then tighten your sender reputation.
If your deal sizes are under $50K, you probably don't need ZoomInfo-level data for biotech prospecting. You need 200 verified contacts per month and a domain that isn't on fire. That's a fundamentally different problem - and a much cheaper one to solve. (If you’re evaluating vendors, compare options in email list providers.)
Go Multi-Channel
Email alone isn't enough. Multi-channel outreach delivers 75% better results than single-channel efforts. One counterintuitive finding from Zymewire's research: blank connection requests outperform generic "like-minded professional" notes. Layer email, phone, and social touches on the same timeline - day 1 email, day 3 call, day 5 connection request. To operationalize this, build a consistent cold calling system alongside your sequences.
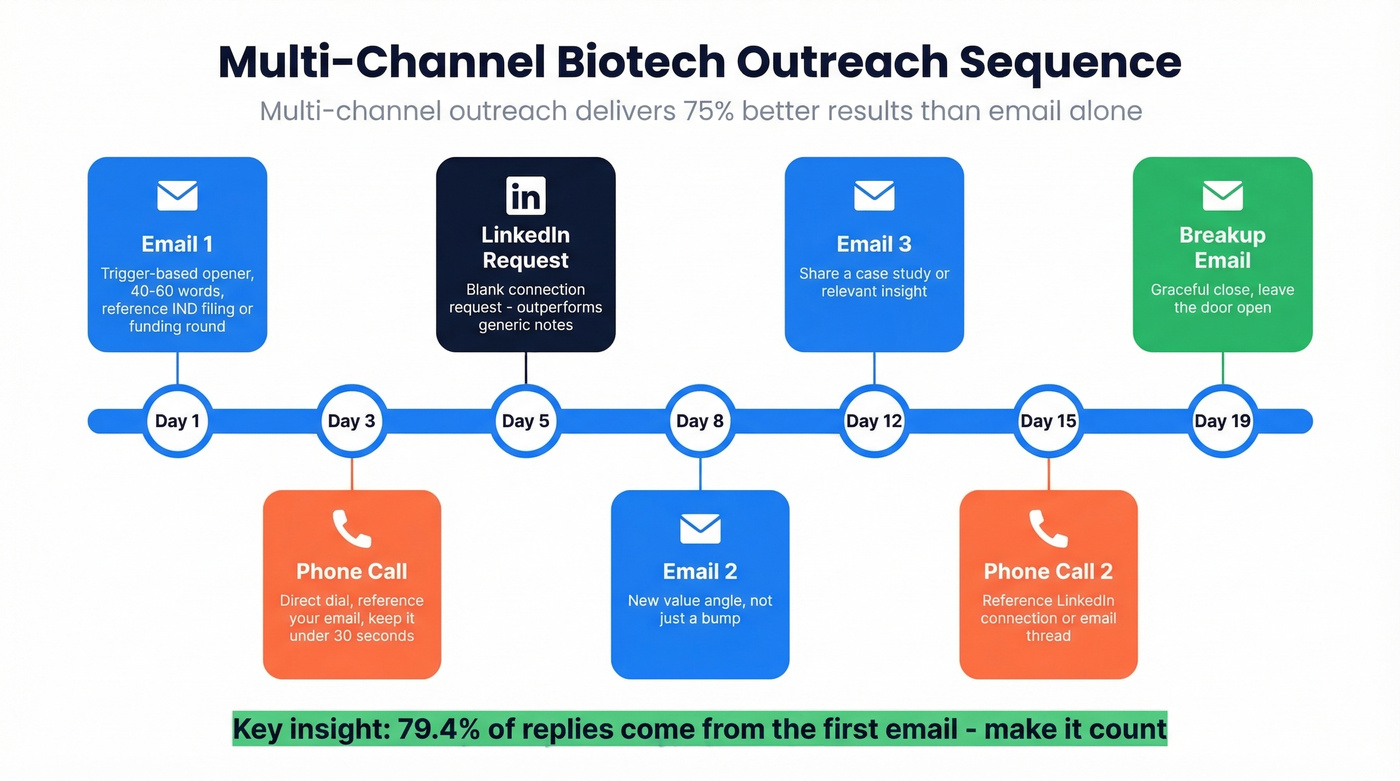
Let's be honest: if you're only sending emails and wondering why biotech VPs aren't responding, you're fighting with one hand tied behind your back. The teams we see booking consistent meetings are the ones combining verified email with direct dials and social touches in a coordinated sequence.
Compliance: What's Actually Legal
Myth: HIPAA prohibits cold emailing biotech professionals. Reality: HIPAA governs patient PHI - protected health information - not professional contact data. A VP of CMC's work email isn't patient data.
CAN-SPAM doesn't require prior consent for B2B email. You can legally cold email biotech professionals in the US as long as you include a valid physical address, a functioning opt-out mechanism, and honest subject lines. That's it.
But don't assume US rules apply everywhere. GDPR covers EU contacts regardless of where you're located. You'll need a legitimate interest basis for B2B outreach and must honor opt-outs promptly. If you're emailing biotech professionals in Basel or Cambridge (UK), GDPR applies to you - skip this at your own risk.
Best Tools for Biotech Outreach
Prospeo
The strongest option for biotech outreach teams that need verified contacts without the enterprise procurement headache. The platform covers 143M+ verified emails at 98% accuracy with a dedicated biotech contact database, 30+ search filters, and buyer intent signals powered by Bombora. The 7-day data refresh cycle is the real differentiator - most list vendors refresh quarterly at best. At roughly $0.01 per email with a free tier of 75 emails/month, you can test before committing. Compare that to traditional list vendors charging $0.20-$0.60 per contact for staler data. If you’re building a repeatable outbound motion, pair this with a clean lead generation workflow.
Apollo.io

Good entry point for smaller teams. Free tier available, paid plans from roughly $49-99/mo per user. Solid built-in sequencing, but less specialized for healthcare or biotech intent signals compared to platforms with dedicated life sciences filtering. If you're running a lean operation and don't need deep biotech-specific data, it's a reasonable starting point.
ZoomInfo
Deep enterprise data and the broadest US database, but typically $15-40K/year with annual contracts. If you just need verified biotech contacts and don't need ZoomInfo's full GTM suite, you're overpaying significantly. Skip this unless your org already has a contract or your deal sizes justify the spend.
| Tool | Verified Biotech Emails | Data Refresh | Starting Price | Contract Required |
|---|---|---|---|---|
| Prospeo | 143M+ (98% accuracy) | 7 days | Free / ~$0.01/email | No |
| Apollo.io | Broad B2B coverage | Standard cadence | Free / ~$49/mo | No |
| ZoomInfo | Deep US coverage | Standard cadence | ~$15K/year | Yes (annual) |

You don't need ZoomInfo pricing to reach biotech decision-makers. Prospeo gives you 143M+ verified emails, 125M+ direct dials, and buyer intent across 15,000 topics - at $0.01 per email. Layer funding rounds, headcount growth, and technographics with 30+ filters built for targeted biotech prospecting.
Enterprise biotech data without the enterprise contract. Start for free today.
FAQ
What reply rate should I expect from biotech cold email?
Plan for roughly 3.58% with about 5 positive responses per 1,000 contacts. Top-quartile campaigns with tight targeting and verified data consistently push above 5.5%.
Is it legal to cold email biotech professionals?
Yes, in the US. CAN-SPAM allows unsolicited B2B email as long as you include a physical address, opt-out link, and honest subject lines. HIPAA governs patient data, not professional contacts. For EU prospects, you need a GDPR-compliant legitimate interest basis.
How does biopharmaceutical outreach differ from general cold email?
Biopharmaceutical outreach requires domain expertise that generic agencies lack - understanding clinical-stage triggers, regulatory timelines, and multi-stakeholder buying committees unique to biopharma. The best campaigns pair verified life sciences contact data with messaging that references real pipeline events like IND filings or Phase II readouts.